|
Navigation
|
Etudiantes: Tatiana Benavides et Line Aschwanden -------------------------------------------------------------------------------------------------------------------------------------------------------------------------------- La méthode de recherche documentaire semble bien acquis! 2/2 A. les mots-clés, domaines, sous-sujets sont bien complets et correctement hiérarchisés 1/2 B. On regrette que les riches mots-clés trouvés n'aient pas été l'objet d'équations plus riches, du style:
2/2 C. Bonne exploration de plusieurs outils de recherche complémentaires (catalogue NEBIS, bases de données bibliographiques, moteurs de recherche) 2/2 D. Les références sont correctes 2/2 E. Le texte répond ŕ la question, mais quelques affirmations ne sont pas référencées. Les réponses aux questions sont appuyées par des informations fiables.Les affirmations dans la partie de réponses aux questions posées sont référencées, c'est bien. 2/2 F. La restitution écrite de la démarche de recherche d'information est bonne: on comprend bien la méthode de recherche documentaire Pour information, on peut utiliser des textes intégraux avec guillemets et source, ou des idées empruntées avec paraphrase et source (droit d'auteur). Mais la reproduction par copié collé d'image, de tableau, de dessin, de graphique, de statistiques, n'est pas possible, męme en citant la source! Pour une reproduction, il faut demander aux auteurs et ŕ l'éditeur la permission de reproduction (droit de diffusiont). Parfois, il faut payer pour obtenir ce droit. Autrement dit, le droit d'utilisation des "images" n'est pas le męme que les idées sous forme écrite! 11/12 pts -------------------------------------------------------------------------------------------------------------------------------------------------------------------------------- 2.1 Mots-clés, synonymes, du plus général (domaine) au particulier Mots-clés: Domaines: Sujets: Sous-sujets:
First, our aim was to understand the general concepts of encapsulation. We searched on Grand dictionnaire terminologique for encapsulation in order to get a basic definition of the word in French as well as to obtain some synonymes (e.g. entrapment) in English. We also looked in Wikipedia (using both languages French and English) for encapsulation, to get also a general idea on the subject, but there were no relevant results. Next, we searched in Google for cell encapsulation, looking for general information, and we found one good article from reliable source: Nature (Ref 1). Then, we started investigating in some general reviews because the topic we chose is very broad. So, we tested our equations in different Bibliographic Data Bases, such as Scopus, ISI Web of Science and PubMed: Afterwards, we searched in a Monograph Catalog, NEBIS, for cell encapsulation. Now we got only 17 results, within a thesis that is actual, relevant and interesting [Ref 7] and a book [Ref 8] that is a good reference but it is not very actual. We also looked into a Publisher BDB, ScienceDirect, again for cell encapsulation, but this time there were too many results (1162)! We noticed though, that the relevant articles were already found with the previously used BDB. As there is a very broad and general view on the technical aspects of cell encapsulation and cell types, all depending on the diversity of the approaches that want to be treated with this technology, we decided to specialize our search within the topic of encapsulation of pancreatic cells encapsulated in PEG. Finally, we searched detailed articles about ONE type of cell (pancreatic cells) with ONE type of encapsulation (PEG = polyethylene glycol). We used the ISI Web of Knowledge (to test another source of reference) and investigated for Equation 2, limiting the search to the Title only. We found one relevant article (Ref 9) and one patent (Ref 10) that helped complementing this research task.
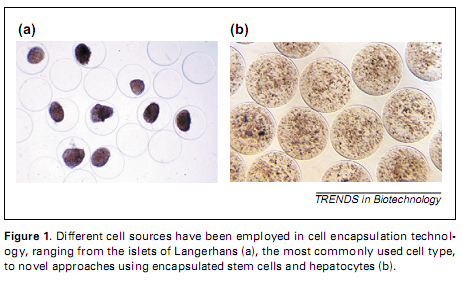
1) ORIVE Gorka, HERNANDEZ Rosa Maria, GASCON R. Alicia, CALAFIORE Riccardo, CHANG M. S. Thomas, DE VOS Paul, HORTELANO Gonzalo, HUNKELER David, LACIK Igor, SHAPIRO A. M. James, PEDRAZ José Luis. Cell encapsulation: Promise and progress. Nature Medicine, 2003, vol. 9, pp. 104-107. 2) ORIVE Gorka, HERNANDEZ Rosa Maria, GASCON R Alicia, IGARTUA Manoli, PEDRAZ José Luis . Encapsulated cell technology: From research to market. Trends in biotechnology, 2002, vol. 20, iss. 9, pp. 382 -387. 3) COLTON K. Clark. Engineering challenges in cell-encapsulation technology. Trends in biotechnology, 1996, vol. 14, iss. 5, pp. 158 -162. 4) ORIVE Gorka, HERNANDEZ Rose Maria, GASCON R. Alicia, CALAFIORE Riccardo, CHANG M.S. Thomas. DE VOS Paul, HORTELANO Gonzalo. HUNKELER David, LACIK Igor, PEDRAZ José Luis. History, challenges and perspectives of cell microencapsulation.Trends in biotechnology, 2004, vol. 22, iss. 2, pp. 87-92. 5) LUKAS J, FENCLOVA T, MOKRY J, KARBANOVA J. Chemistry, physical properties and biocompatibility of hydrogels for immunoprotection of mammalian cells. Chemicke Listy, 2004, vol. 98, iss. 1, pp. 14-21. 6)HUNKELER D, RAJOTTE R, GREY D, MOREL Ph, SKJAK-BREAK G, KORBUTT G, GILL R, OBERHOLZER J. Bioartificial Organ Grafts: A View at the Beginning of the Third Millennium. Artificial cells, blood substitutes, and immobilization biotechnology, 2005, vol. 31, iss. 4, pp. 365 . 7)CELLESI Francesco. New fully synthetic materials for cell encapsulation. Thčse. Zürich: Eidgenössische Technische Hochschule ETH Zürich, 2003, 131 p. 8)KÜHTREIBER M. Willem, LANZA P. Robert, CHICK L. William. Cell encapsulation technology and therapeutics. Boston: Birkhäuser, 1999. 9)WEBER LM, CHEUNG CY, ANSETH KS. Multifunctional pancreatic islet encapsulation barriers achieved via multilayer PEG hydrogels. Cell transplantation, 2007, vol. 16, iss. 10, pp. 1049-1057. 10)KIZILEL Seda, WYMAN L Jason, MRKSICH Milan, NAGEL R Sidney, GARFINKEL R Marc. Microencapsulation of pancreatic islets within GLP-1 functionalized poly(ethylene glycol) (PEG) hydrogel using selective withdrawal. Brevet US 2007/0190036 A1. 3 janvier 2007. Attention: ŕ la fin d'une référence, mentionnez l'outils de recherche qui l'a trouvé! 2.3 Réponse aux questions: 1) quelles cellules encapsuler? 2) dans quelle capsule? 3) what about pancreatic cells inside a PEG capsule? In this section, we respond to our two questions in the general domain, in additon to our specific question where we focus on pancreatic cells and PEG encapsulation. We mention the reference where we took the information from (paraphrasing text and images), and include also a few direct citations (in quotation marks) from some sources. 1) quelles cellules encapsuler? Practically, any cell can be encapsulated, depending on the approach we want to treat, that in most cases would be curing a disease such as diabetes or cancer. These cell types have already been encapsulated: kidney cells, BHK cells, C2C12 cells, diabetes islets, pig liver cells and human immortalized cell line. [Orive et al., 2002] The following cell types are also commonly used for encapsulation: fibroblasts, myoblasts, kidney cells, pancreatic islets, ovary cells, parathyroid cells, hepatocytes, chondrocytes, leydig cells, adrenal chromaffin cells, stem cells, PC12 pheochromocytoma cells, myeloma cells, hybridoma cells, tumor cells, virus producer cells and bacteria [Orive et al., 2004] From this reference we can see two examples of encapsulated cells in the figure below:
An important approach is the artificial organ research, where isolated or immunoprotected transplanted cells are being encapsulated. Such cells are predominantly hepatocytes, islets, keratinocytes and chondrocytes. It is important to note the potential each cell type has, for example: stem cells can differentiate in culture into a variety of cell lineages; cell lines can be reversibly immortalized, while maintaining their specific characteristics; neonatal xenogeneic tissue is less expensive to isolate and is also expandable in culture; and cryopreservation of tissue for banking can be obtained in high yield. For bioartifical organs, allografts done with genetically modified human tissue, hold a future promise, compared to xenografts. Human tissue is derived from cadavers with the potential of immortalized cells lines based on pluripotent stem cell technology. Bioartificial cartilage, skin and bone belong to a second category of bioartificial organs where cells are seeded to an often biodegradable substrate. Another relevant approach where encapsulated cells are used is specifically in gene therapy. Here, the transfection of islets with immunomodulatory genes such as Fas L and CTLA4-IG has been done, as well as antiapoptotic genes such as bcl-2. [Hunkeler et al., 2005]
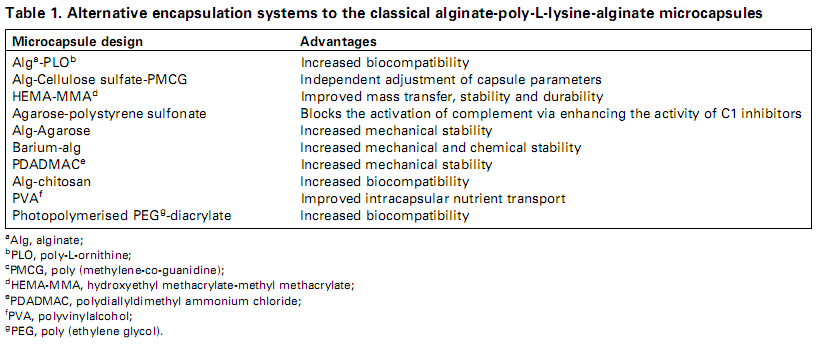
Some common biomaterials used to encapsulate cells include: sodium alginate, alginate-PLL capsules and hollow fibers. The advantage on using alginate is that it has low endotoxin content, while Poly-L-lysine (PLL) has reduced membrane porosity. [Orive et al., 2002] Other encapsulating biomaterias are also: HEMA-MMAa, agarose-polystyrene/sulfonic acid, AN69, cellulose sulfate, acetate, alginate-agarose and chitosan. [Orive et al., 2004] The following table was obtained from this reference, and it helps illustrate the combination of biomaterials used to design a microcapsule according to their advantages:
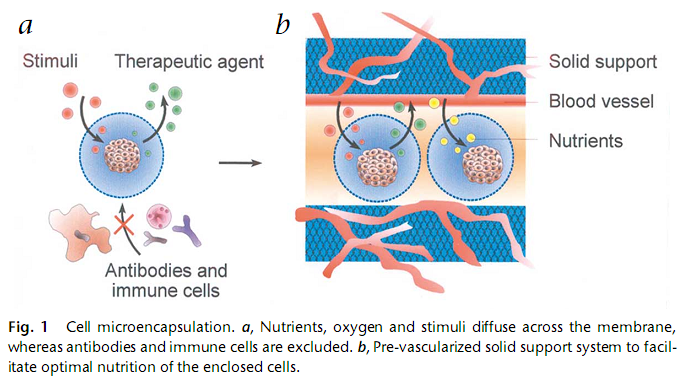
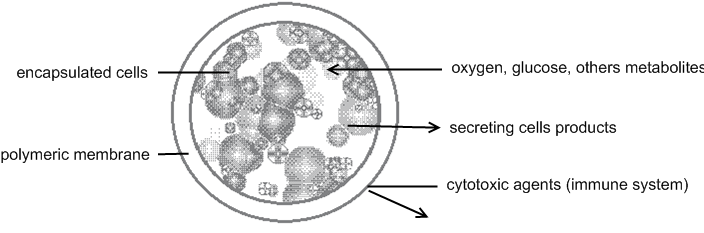
Polymer hydrogels form a semipermeable membrane around the cells, permiting diffusion of oxygen and necessary metabolites, while secreting cell products and restricting the entrance of large cytotoxic agents from the immune system. [Lukas et al., 2005] From the same reference, there is a picture to illustrate the principle of encapsulation :
A promising approach of using encapsulated cells, as mentioned before, is in the implementation of bioartifical organ grafts. In this case, co-encapsulation and immune protection provide a better humoral hormonal response and graft survival. From [Hunkeler et al., 2005] we have a very clear definition of what a bioartifical organ represents, it is "a transplantable device providing a mechanically stable biocompatible, non-degradable host to living cells. Examples include macro- and microencapsulated cells used for immunoisolation such as the bioartificial pancreas (islets), liver (hepatocytes), and parathyroid (thyroid) [...] Overall, bioartificial organs are comprised of cells immunoisolated by elastic polymers or semi-permeable membranes derived from synthetic and naturally occurring products such as polysaccharides." Alginate beads and PEG coating give promising results as encapsulation techniques, due to the fact that PEG creates a camouflage surface against the immune system. Besides, polyelectrolytes cause a minimal host response. An advantageous technique in this domain is the coencapsulation of different biomaterials to enhance their properties, as to provide local immunosuppression. [Hunkeler et al., 2005] 3) what about pancreatic cells inside a PEG capsule?
Mots-clés:
islet encapsulation, multilayer hydrogel, immunoprotection, cell-matrix interactions, pancrea(*), beta cell(s), PEG, poly(ethylene glycol), capsules, diabetes, insulin Information about this type of encapsultaion : Pancreatic islets embedded in an hydrogel of poly (ethylene glycol) (PEG): a sequential photopolymerization PEG hydrogel is used to create many layers of hydrogels which have different isolation functions (for immunoprotection). PEG hydrogels are bioinert, meaning no interactions between PEG and cells, because it iso full of water that there is slow protein adsorption. The protocol to make this type of encapsulation is described in [Weber et al., 2007]. The following picture which comes from the same reference, explains how to create a two-layered hydrogel (photopolymerization of dimethylacrylated PEG in Teflon molds) : 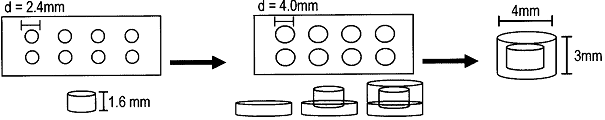 Simple stepwise depiction of two-layer PEG hydrogel fabrication And here you can see cells inside the capsule (same reference) : 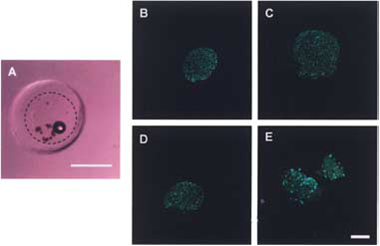 Digital image looking down at islets encapsulated within the interior layer of a two-layer hydrogel, with scale bar representing 2 mm and dashed line indicating the outline of the interior layer (A), and LIVE/DEAD staining for islet viability after 1 day (B), 7 days (C), 14 days (D), and 28 days (E) in culture within two-layer PEG hydrogels. Live cells are identified by red fluorescence. Scale bar: 100 ěm (B–E).
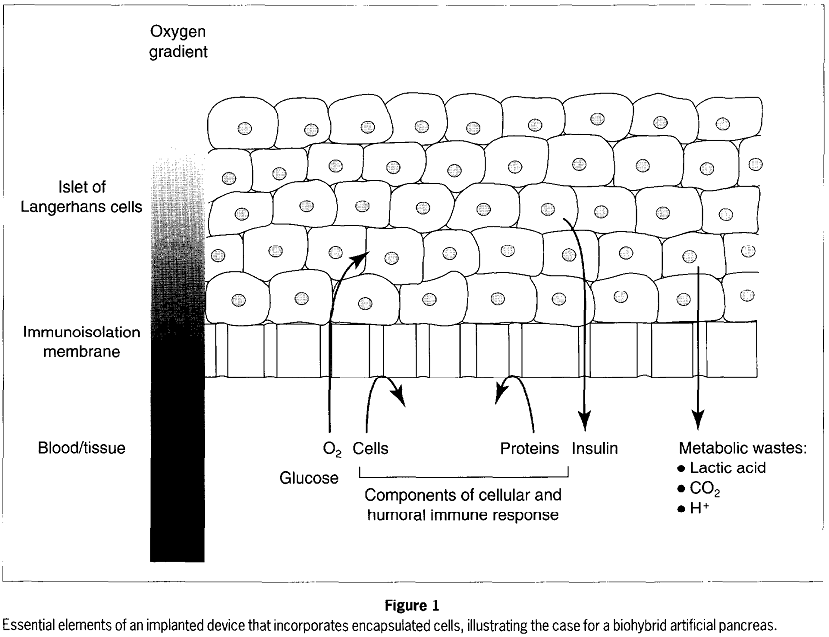
The PEG can be functionalized coating an insulinotropic agent (glucagon-like peptide 1 (GLP-1)). All indications on how to produce this capsule are given in [Kizilel et al., 2007]. From [Colton et al., 1996], we have the following illustration of a biohybrid artificial pancreas:
|